- Sign up for our Newsletter!
- +49 (0)2151 - 8777-0
- info@calorplast.de
- Mon - Fri: 8:00 - 17:00
Redox-Flow Battery: The Forward-Looking Solution for Efficient Energy Storage?
You are here:
- Home
- Redox-Flow Battery: The Forwar…
In a world where sustainable and efficient energy storage is becoming increasingly important, the redox-flow battery is gaining momentum among future-oriented companies. In particular, the vanadium redox-flow battery offers a powerful alternative to conventional battery technologies. But how exactly does this system work, and what advantages does it offer for industrial applications?
The key to the high efficiency of this technology lies in the unique operating principle of the redox-flow battery: Energy is not stored in solid electrodes but in liquid electrolytes that circulate through separate tanks. This enables the independent scaling of capacity and power – a decisive benefit for companies that need to store and access large amounts of energy flexibly.
Another important aspect is the thermodynamics inside the battery. The use of a redox-flow battery heat exchanger optimizes temperature management and increases the lifespan as well as the performance of the storage system. This is particularly relevant in high-performance applications, where maintaining a constant operating temperature is essential for efficiency and safety.
What Is a Redox-Flow Battery?
A redox-flow battery is an electrochemical energy storage system based on the conversion of chemical energy into electrical energy. Unlike conventional batteries, where energy is stored in solid electrodes, redox-flow batteries use liquid electrolytes that circulate in external tanks. These liquids contain dissolved active materials that undergo chemical reactions at special electrodes inside the cell to supply or absorb electrical energy.
A particular advantage of this technology is the flexibility of capacity scaling: While the power output of the battery is determined by the size of the electrodes, the storage capacity can be scaled by the volume of the electrolyte tanks. This makes vanadium redox-flow batteries especially attractive for industrial applications requiring efficient storage and on-demand retrieval of large amounts of energy.
Moreover, the operation of a redox-flow battery is characterized by long service life and high cycle durability, as the electrochemical reactions take place within the liquid electrolytes, avoiding mechanical or structural degradation of solid electrodes. This significantly reduces maintenance and operating costs. Another key element for reliable performance is thermal management. A redox-flow battery heat exchanger keeps the temperature stable, ensuring optimal efficiency and extended battery lifetime.
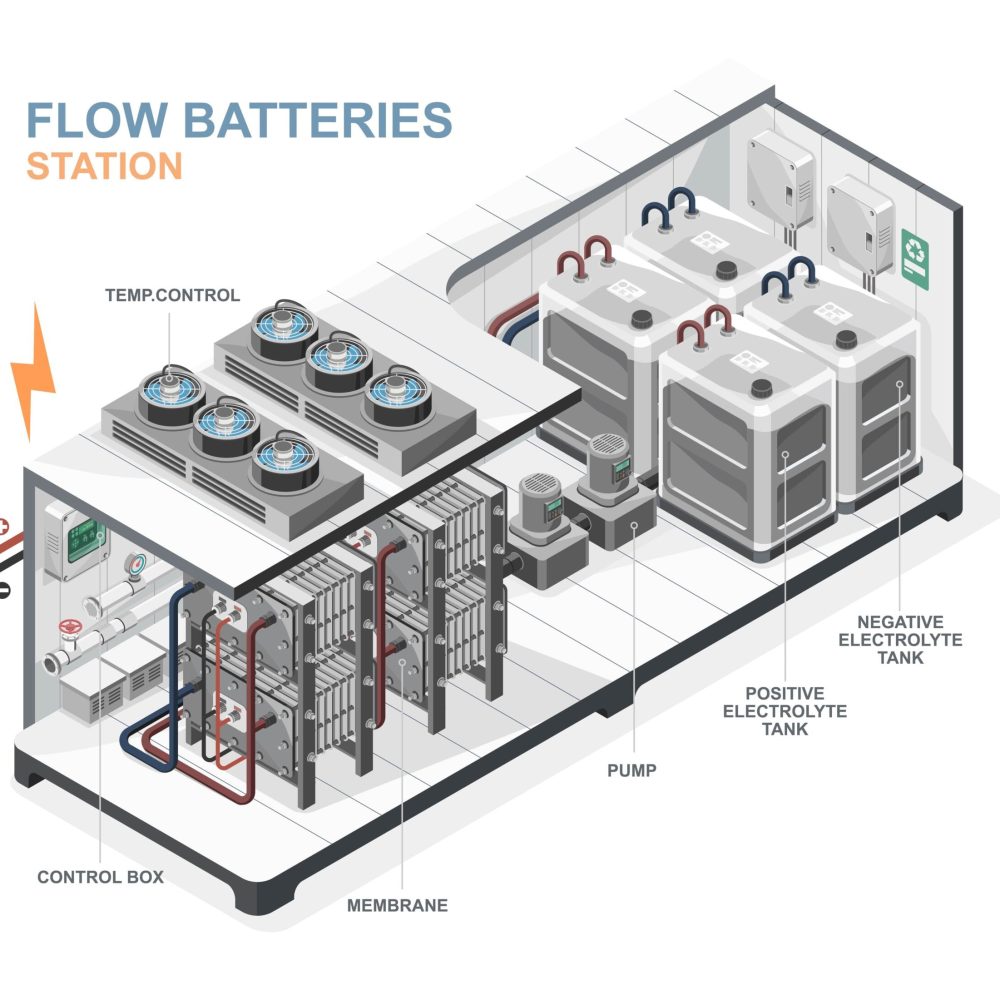
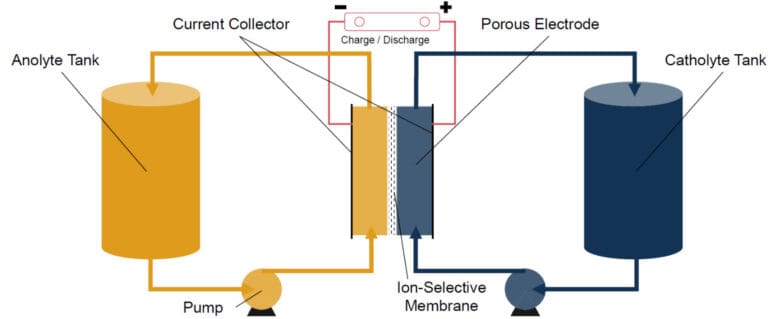
Structure of a Redox-Flow Battery
A redox-flow battery consists of several essential components that together enable efficient energy storage. The core elements are the two electrolyte tanks, each containing ion-rich liquids – often based on vanadium. These liquids circulate in separate loops through the electrochemical cell, where the energy conversion takes place.
The electrochemical cell itself consists of two half-cells separated by an ion-conducting membrane. This membrane allows controlled ion exchange between the electrolytes while electrons flow through an external circuit to generate usable electrical energy. The battery’s power is determined by the size of the cell stack, while storage capacity is scaled by the volume of the electrolyte tanks – a major advantage compared to conventional batteries.
Because charging and discharging generate heat, efficient thermal management is essential. A specially designed redox-flow battery heat exchanger ensures stable temperatures and minimizes thermal losses, which significantly contributes to system longevity and efficiency.
Due to this modular design, redox-flow batteries offer flexible and scalable solutions for industrial applications. Vanadium redox-flow systems, in particular, impress with high cycle stability, low degradation, and long service life – ideal for economic and sustainable energy storage.

How a Redox-Flow Battery Works
The operating principle of a redox-flow battery is based on electrochemical reactions occurring in liquid electrolytes. Unlike conventional batteries, this system enables flexible scaling of power and capacity – ideal for industrial energy storage requiring high efficiency and durability.
How does a redox-flow battery work?
Two liquid electrolytes circulate through separate tanks and react within an electrochemical cell. A membrane separates the half-cells, while electrons flow through an external circuit to generate electrical energy. Redox-flow batteries benefit from a dedicated heat exchanger that thermally stabilizes the system and boosts overall efficiency.
How long do redox-flow batteries last?
Redox-flow batteries offer an extremely long service life of over 20 years and more than 10,000 charge cycles. Since no solid electrodes are subject to wear, the capacity remains stable over the long term. A redox-flow battery heat exchanger optimizes the operating temperature and contributes to the efficiency and longevity of the system.
Why is the correct temperature level important?
A stable temperature level is essential for the operation of a redox-flow battery, as extreme temperatures can affect the electrochemical reactions and reduce the system’s efficiency. Particularly in vanadium redox-flow batteries, the operating temperature must be kept within an optimal range to maintain the viscosity of the electrolytes and ensure consistent performance.
This is where the redox-flow battery heat exchanger comes into play: it provides precise temperature regulation, prevents overheating, and minimizes energy losses. Efficient thermal control not only extends the service life of the redox-flow battery but also optimizes its charging and discharging processes—an essential factor for industrial applications.
Why is the correct selection of the heat exchanger so important for redox-flow batteries?
Choosing the right redox-flow battery heat exchanger is essential to ensure a stable operating temperature and maintain high system efficiency. Since the electrochemical processes in a redox-flow battery are highly temperature-dependent, insufficient heat dissipation can lead to performance losses or accelerated degradation of the electrolytes.
Plastic heat exchangers offer key advantages over metallic heat exchangers, particularly in chemically demanding applications such as redox-flow batteries. The plastics used by Calorplast in their heat exchangers are characterized by excellent chemical resistance. They are resistant to a wide range of aggressive media, which ensures an exceptionally long service life even under corrosive conditions. In contrast to metal heat exchangers, plastic heat exchangers do not affect the electrolyte, as they do not release metal ions into the medium. This is especially important in electrochemical processes, where foreign ions could negatively impact the functionality and longevity of the electrolyte. Thanks to the modular design of our Calorplast heat exchangers, they are—just like the redox-flow battery itself—scalable. They can be adapted easily and flexibly to different performance requirements, whether for small laboratory systems or large-scale industrial applications.
A heat exchanger specifically tailored to vanadium redox-flow batteries ensures precise temperature control and prevents overheating or unwanted temperature fluctuations. As a result, the battery remains functional over the long term, the efficiency of charge and discharge cycles is optimized, and the overall service life of the storage system is significantly extended—an essential factor for industrial applications with high demands for reliability and cost-effectiveness.
Depending on the system design and thermal control requirements, the heat exchanger can be installed either directly inside the electrolyte tank or in the feed line before the electrochemical cell.
Types of Redox-Flow Batteries: An Overview
Depending on the electrolytes used and the technical design, different types of redox-flow batteries exist, suited for various industrial applications. The main variants include:
Vanadium Redox-Flow Battery (VRFB)
The most widely used technology relies on vanadium electrolytes in two separate tanks. This prevents electrolyte cross-contamination and ensures exceptional cycle stability. VRFBs offer long service life and are ideal for stationary energy storage in industrial and utility sectors.
Zinc-Bromine Redox-Flow Battery
This battery type uses zinc and bromine solutions. It features high energy density but requires precise temperature control. Here, a heat exchanger plays a crucial role in maintaining electrochemical stability and overall efficiency.
Iron-Chromium Redox-Flow Battery
Using iron and chromium ions as active materials, this is a cost-effective alternative but offers lower energy density. Due to its robust construction, it is preferred for large-scale systems requiring many charge cycles.
Hybrid Redox-Flow Battery
This variant combines traditional redox-flow technology with solid electrode materials to increase energy density. While more compact, it places higher demands on material selection and thermal regulation.
Each of these redox flow batteries has specific advantages and challenges. The selection of the appropriate storage type depends on individual requirements for capacity, cost, and thermal stability.
Applications and Use Cases of Redox-Flow Batteries
Redox-flow batteries offer numerous advantages for industrial and commercial applications, particularly due to their high cycle stability and scalable capacity. Key application areas include:
Energy storage for renewable energies
Wind and solar farms benefit from redox-flow batteries, as these systems store excess energy and release it when needed. Thanks to their long service life and flexible scalability, they are a sustainable solution for grid stabilization.
Industrial operations with high energy demand
Companies with energy-intensive processes—such as those in the glass, metal, or chemical industries—use vanadium redox-flow batteries to reduce peak loads and lower operating costs.
Backup power supply and grid stabilization
Redox-flow batteries can serve as a reliable backup solution for critical infrastructure such as data centers, hospitals, and telecommunications facilities. They ensure an uninterrupted power supply without performance loss.
Use in microgrids and island solutions
In remote regions or island grids, vanadium redox-flow batteries enable independent energy supply by efficiently storing renewable energy and increasing grid autonomy.
Thermal management in battery systems
An efficient redox-flow battery heat exchanger is essential for maintaining a constant operating temperature and maximizing battery lifetime. In large-scale installations, precise
Feel free to take a look at our case study on the use of redox-flow batteries and see for yourself based on a real-world example.
Advantages and Disadvantages of Redox-Flow Batteries
Redox flow batteries have established themselves as high-performance energy storage devices for industrial applications. Especially when combined with renewable energy sources, they offer a reliable and long-lasting solution. However, as with any technology, there are both advantages and challenges.
Advantages:
Long service life
Since the electrochemical process in redox-flow batteries operates with virtually no material wear, they achieve a high number of charge cycles without significant capacity loss.
Independent scalability of power and capacity
The storage capacity can be increased by adjusting the amount of electrolyte liquid, while the power output is determined by the size of the electrode surface. This allows flexible adaptation to different requirements.
Safe technology
Vanadium redox-flow batteries are non-flammable and operate at low operating temperatures, minimizing the risk of thermal overheating.
Ideal for grid-stabilizing applications
Due to their highly stable charging and discharging behavior, redox-flow batteries are ideal for grid stabilization and the storage of renewable energy.
Efficient thermal management
The use of a redox-flow battery heat exchanger enables precise temperature control, optimizing both system performance and lifespan.
Self-sufficient redox-flow storage system
A portion of the generated electrical energy is used for the system’s own operation, making it independent of external power supply. This allows reliable and efficient operation of essential system components such as circulation pumps that maintain continuous electrolyte flow between the tanks and the electrochemical cell. Cooling units or other temperature-regulating systems used to cool the electrolytes via integrated heat exchangers can also be powered by the energy generated internally.
Disadvantages:
Comparatively large design
Compared to lithium-ion batteries, redox-flow storage systems require more space because they use tanks to store the electrolyte fluids.
Lower energy density
Redox-flow batteries have a lower energy density compared to conventional battery technologies, making them more suitable for stationary rather than mobile applications.
Higher upfront investment costs
Initial investment costs are higher than for other battery systems because the electrolytes represent the most expensive and valuable component of vanadium redox-flow batteries. In return, they significantly determine the system’s return on investment (ROI). To maximize their service life, the use of a heat exchanger is essential, as it ensures the thermal stability of the system.
Electrochemical reactions are efficient and stable only within a specific temperature range
During charging and discharging, redox reactions occur within the battery’s half-cells. These chemical processes are not fully efficient — part of the input energy is released as heat. Especially at high current density and in large-scale systems, the electrolyte temperature rises rapidly. If this defined temperature range is exceeded, efficiency decreases, side reactions increase, and irreversible damage may occur.
Despite these challenges, redox-flow batteries remain a future-proof solution for many industrial applications, especially when sustainable and durable energy storage is required.